Microsilica is a highly reactive pozzolanic powder produced during the manufacture of ferrosilicon and silicon metal alloys.
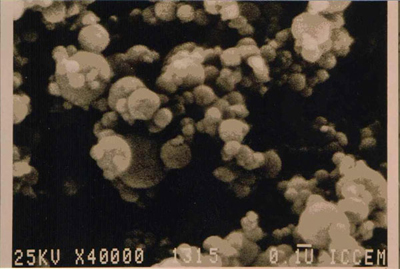
 Microsilica powder is composed of ultra fine solid spherical particles of amorphous silica each approximately 100 times smaller than a cement grain.
Microsilica powder is composed of ultra fine solid spherical particles of amorphous silica each approximately 100 times smaller than a cement grain.
It is formed by the condensation of vapour during the smelting of coal, quartz, iron ore and other materials in an electric arc furnace at around 2000. The vapour and furnace gasses travel through a heat exchange system at the end of which the by now solid particles of amorphous silica are separated from the gases via a bag filter system.
Once captured it goes through an aeration process to increase its bulk density to facilitate handling and also to reduce the unit cost of transport.
Microsilica performs two basic functions in hardening and hardened concrete. It is a reagent, reacting with calcium hydroxide to produce calcium silicate hydrates to supplement those already resulting from the mixing of cement and water, increasing concrete strength.
It also acts as a pore filler to reduce the ingress of airborne and waterborne deleterious gases and chemicals, so protecting the steel reinforcement within.
There are many other advantages. For further information see the microsilica performance section.
Products
Further Information
There are many different ways in which our products can benefit from concrete performance.
Click on the tabs below to find out how.
-
Strength
-
The Reaction
MS 93 concrete shows increased strength throughout the curing cycle.
This is especially useful in any application where concrete with a high early strength is required so that day to day activity is not interrupted for any great length of time.
The hydration of OPC produces many compounds, including calcium silica hydrates and calcium hydroxide. The hydrates are the source of strength in concrete.When microsilica is added to fresh concrete it reacts with the calcium hydroxide to produce extra hydrates and so additional strength.
High early strength
- taxiways
- runways
- aircraft standing aprons
- distribution centre roadways and bays
- factory floors
- machinery anchoring
- warehouse flooring
- grouts
- shotcreteGeneral high strength
- structural load bearing applications such as beams
- mining concrete
- core concrete in high buildings
- piling
- foundations
- sea defences
- crash barriers
- shotcrete
-
-
Chemical resistance
-
MS 93 acts as a pore filler greatly reducing the ingress of harmful chemicals and the subsequent corrosion / carbonation of steel reinforcement.
- piling concrete that can be subject to attack by chlorides or sulphates etc
- sea defences
- general marine concrete
- areas where road salt is present
- sewage treatment concrete
- agricultural concrete40% of the concrete placed during the development of Dubai contains 5% microsilica. It is also used extensively in the rest of the UAE Oman and Saudi Arabia.
- below ground its function is to protect foundation and piling concrete from chloride attack by seawater and leached chlorides and other chemicals present in the porous limestone.
- above ground it performs the same function against airborne chlorides resulting from evaporation from the sea in high climatic temperatures.
-
-
Abrasion resistance
-
MS 93 concrete will last longer than non microsilica concrete in areas of high abrasion.
- industrial flooring
- warehouse flooring
- sea defences
- runways
- distribution centre roadways
- agricultural concrete
- scrapyards
-
-
Wet phase
-
MS 93 concrete shows very high flow characteristics against non microsilica concrete for equivalent slump.
- difficult access situations such as mining applications
- pumped concrete
- self levelling concrete
- industrial floors
-
-
Freeze thaw
-
MS 93 concrete gives a much improved resistance to freeze / thaw cycles. It should be used in conjunction with aerated concrete.
- any outdoor concrete on which water collects
- roads
- runways
- pavements
- channels
- roofs
- hard standing
-
-
Alkali silica reaction
-
MS 93 reduces hydroxyl ion concentration in pore solutions thus significantly reducing the risk of ASR.
- concrete containing reactive aggregates
-
-
Cohesiveness
-
MS 93 significantly reduces washout during the pouring of underwater concrete.
It will also greatly reduce rebound during the application of spray concrete so reducing waste, and also enable the application of deeper coats thus saving time in application.
- submarine pouring of concrete
- shotcrete applications
-
-
Electrical conductivity
-
MS 93 reduces the electrical conductivity of concrete.
- high voltage industrial areas
- power stations
- aluminium potlines
- foundations for electric arc furnaces
- substation foundations
-
-
Cement replacement
-
MS 93 can replace OPC.
- up to 35% of OPC in a mix at a ratio of up to 4.5 / 1 for equivalent strength.
-
-
Spray concrete
-
Microsilica 93 is widely used in this application for the following reasons.
- Cohesiveness: A thicker coat can be applied with each pass. Saves on application time
- Reduces rebound: Reduces wastage, cleanup cost and time
- Increased density: Protects steel reinforcement from harmful chemicals
- Higher strength: Less concrete required
-
-
Impact resistance
-
High strength and density combine to give a high resistance to impact.
- slipform crash barriers
- warehouse flooring
- heavy industrial flooring
-
-
Bleeding and segregation
-
Bleed water will often raise fine particles to the surface of the fresh concrete.
- this will later become a weak layer on the surface of the concrete. MS 93 will eliminate both bleeding and segregation in fresh concrete due to greatly improved particle packing.
- the elimination of aggregate segregation prevents the gravity migration of aggregates in a static curing mix.
-
-
Curing
-
Best practice normally adhered to when curing non microsilica concrete should also be followed when curing concrete containing MS 93
-
2. Refactory
-
Microsilica in refactories
-
Microsilica is used in both monolithic and shaped refractories for several reasons.
- Microsilica reacts with with alumina forming mullite which increases the tensile strength of the mix at all temperatures
- It enhances green strength
- It acts as a void filler thus reducing permeability
- It increases thermal shock resistanceIn monolithics it also offers advantages similar to those seen during the application of shotcrete/spray concrete in the construction industry.
- Reduction of rebound reducing wastage and cleanup
- Cohesiveness meaning a thicker coat can be applied with each pass thus speeding up application time.
-
3. Other Applications
Casting
Insulation
Non cementitious building products

Microsilica is an amorphous form of silica. It is not crystalline. This important.
Please see our MSDS sheet here
Slurries are sometimes sold on the basis that there is some health and safety issue with powders. This is a nonsense.
Typical Physical Composition
| OPC | Microsilica | GGBFS | PFA | |
| Surface area kg/m2 |
360-500 | 15,000-22,000 | 300-500 | 300-600 |
| Specific gravity | 3.12 | 2.10 | 2.90 | 2.30 |
| Bulk density kg/m2 |
1,300-1,400 | 600 | 1,000-1,200 | 1,000 |
Typical Chemical Composition
| % | OPC | Microsilica | GGBFS | PFA |
| SiO2 | 21 | 90/97 | 38 | 50 |
| AI203 | 5.1 | 0.61 | 11.5 | 28.0 |
| CaO | 63 | 0.39 | 41.5 | 3.0 |
| MgO | 0.1 | 1.58 | 7.3 | 2.0 |
| Fe2O3 | 3.5 | 0.94 | 0.3 | 10 |
Electron micrograph of individual microsilica particles at x 40,000 magnification.
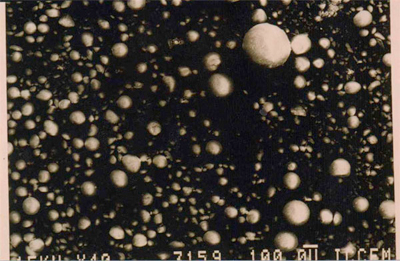
Electron micrograph of densified microsilica agglomerates at x 40 magnification.
The production and history of microsilica
Microsilica has an extraordinary versatility as a high-performance cementitious additive. It is used as a concrete improver or as an eliminator of potentially damaging reactions in certain mixes, and is used extensively in refractories.
Microsilica is the by-product of an unrelated industry, the production of ferroalloys. The raw materials for the production of ferrosilicon, silicon metal and Microsilica are quartz and reducing agents, mainly coal and coke. The overall reaction in the furnace is:
SiO2 (from quartz) + 2C (from coal/coke) = Si and 2CO.
For this reaction much energy is required to produce heat. This is supplied by the arc of the electrodes submerged in the furnace. Iron is also added as a catalyst.
The overall reaction can be divided into several steps. In the highest temperature zone, a cavity beneath the electrode tips, quartz reacts with silicon carbide forming gaseous silicon monoxide and CO and liquid metal which is continuously being tapped from the furnace.
SiO2 + SiC = Si(liquid) + SiO(gas) + CO (gas).
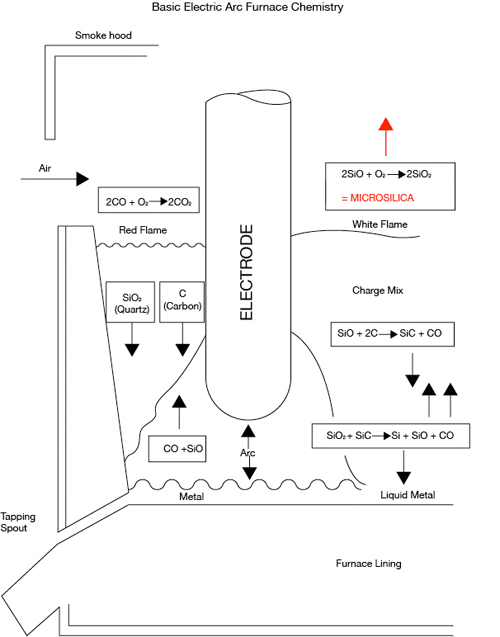
The gases formed in the cavity rise through the charge-mix and react on the way with raw material which is continuously fed to compensate for reacted material. Not all the silicon monoxide reacts with the carbon, some of it escaping the charge and rising to burn in air. Small dusty amorphous particles of about 0.1 micron in size are formed. These are then blown out of the plant with the waste gases.
Attached to each furnace are two powerful electric fans which draw out the gases and smoke, and drive it through filter bags in the baghouse. The bags remove all the dust (microsilica) from the smoke.
The next process involves the micro-pelletisation of the microsilica the forming of the dust into small spheres about 0.5-1mm in diameter. The reasons for this are threefold. Firstly, the bulk density of raw Microsilica, being almost as fine as cigarette smoke, is about 0.2 tonnes per cubic metre. The process of micropelletisation increases the bulk density to between 300 and 650 tonnes per cubic metre. This can reduce transport costs by as much as 65 per cent and so also the final cost to the customer.
Secondly, due to its fineness microsilica is very difficult to handle. This process produces a material with the handling characteristics of fine sand without using a binder, and so avoids the problems that can be caused by foreign materials in the final mix.
Thirdly, due to their shape and size the micropellets move very freely. This factor makes the silo storage, in-plant transfer (mechanical or air-blown) and eventual use simpler than with raw powder. The pellets do not stick to one another, and therefore any fouling and blockage problems are virtually eliminated. With a moderate amount of shear the pellets break up in water and disperse as effectively as raw powder or slurrified material.
These gases were not filtered until the 1970`s, with the introduction of far stricter environmental legislation in many West European countries.
Originally the filtering of this dust brought up the question of whether it could be used to some advantage. In 1975 the IPFEO, the association of ferro-alloy producers in Western Europe, offered 10,000 Swiss francs in reward for a feasible idea as to how the dust could be used.
The prize was awarded to some Danish scientists who proposed the idea of using microsilica in waterglass. As it turned out the use of microsilica in cementitious environments became much more promising.
Initially microsilica was used as an additive in Denmark, Norway, Canada and Iceland. Iceland had suffered for some years from an acute alkali-silica reaction (ASR) problem. The volcanic rock used as aggregate in the concrete is partly glassy and therefore reactive. The Icelandic cement is high in alkalis and in the Reykjavik area the aggregates are dredged from the sea. Furthermore, as a consequence of the geographical location in the middle of the north Atlantic, driving rains are common in Iceland. Therefore, the three parameters governing alkali-silica reaction in concrete: high alkalis, reactive aggregates and high humidity, are all present.
There are different grades of alloy, the production of which give fumes of various grades. The SiO2 content, depending on the type of metal produced, can range between 50 per cent and 98 per cent.



